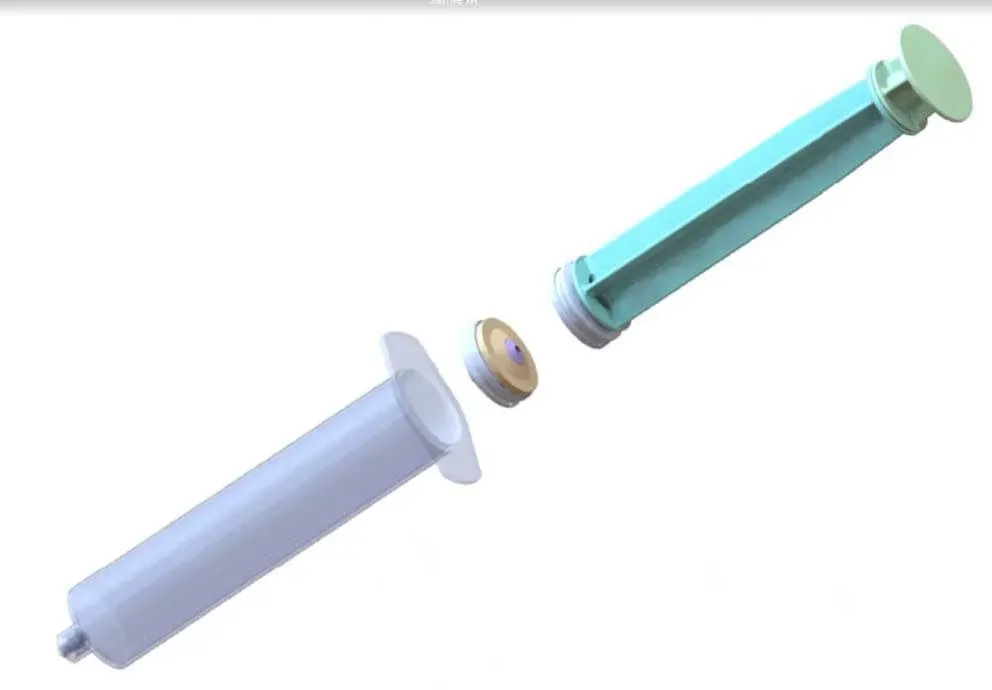
Innovating Intravenous Drug Delivery
Sadleir Laboratories proudly presents IRIS, the test dose syringe designed to transform intravenous drug administration with unmatched precision and safety. Discover how our cutting-edge technology can improve patient care and streamline healthcare practices.
Preventing Intravenous Anaphylaxis
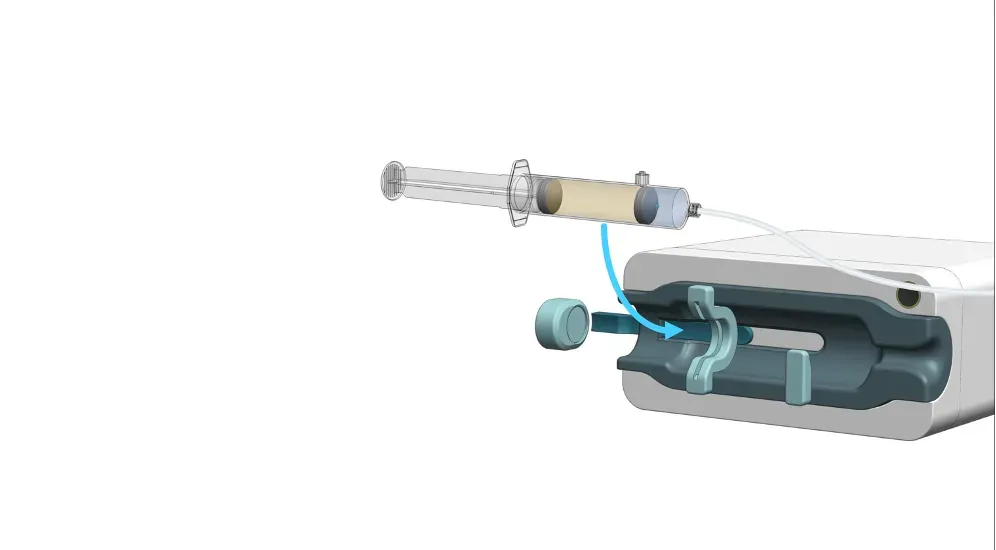
Automatic Intravenous Dosing
Convenient test doses without Interruption of the Therapeutic Dose and without User Intervention
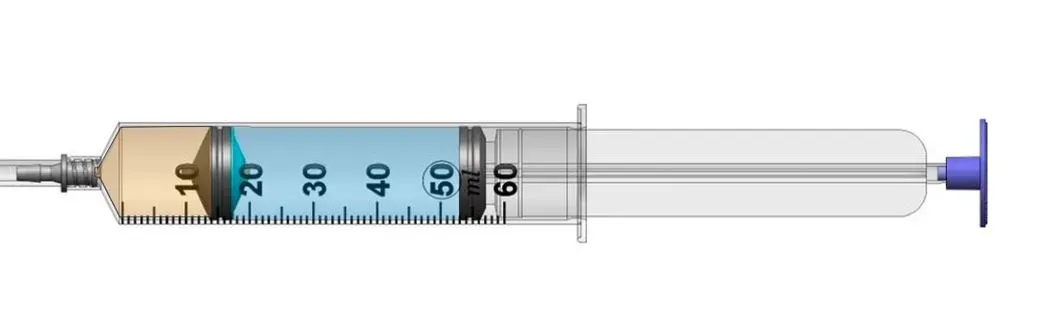
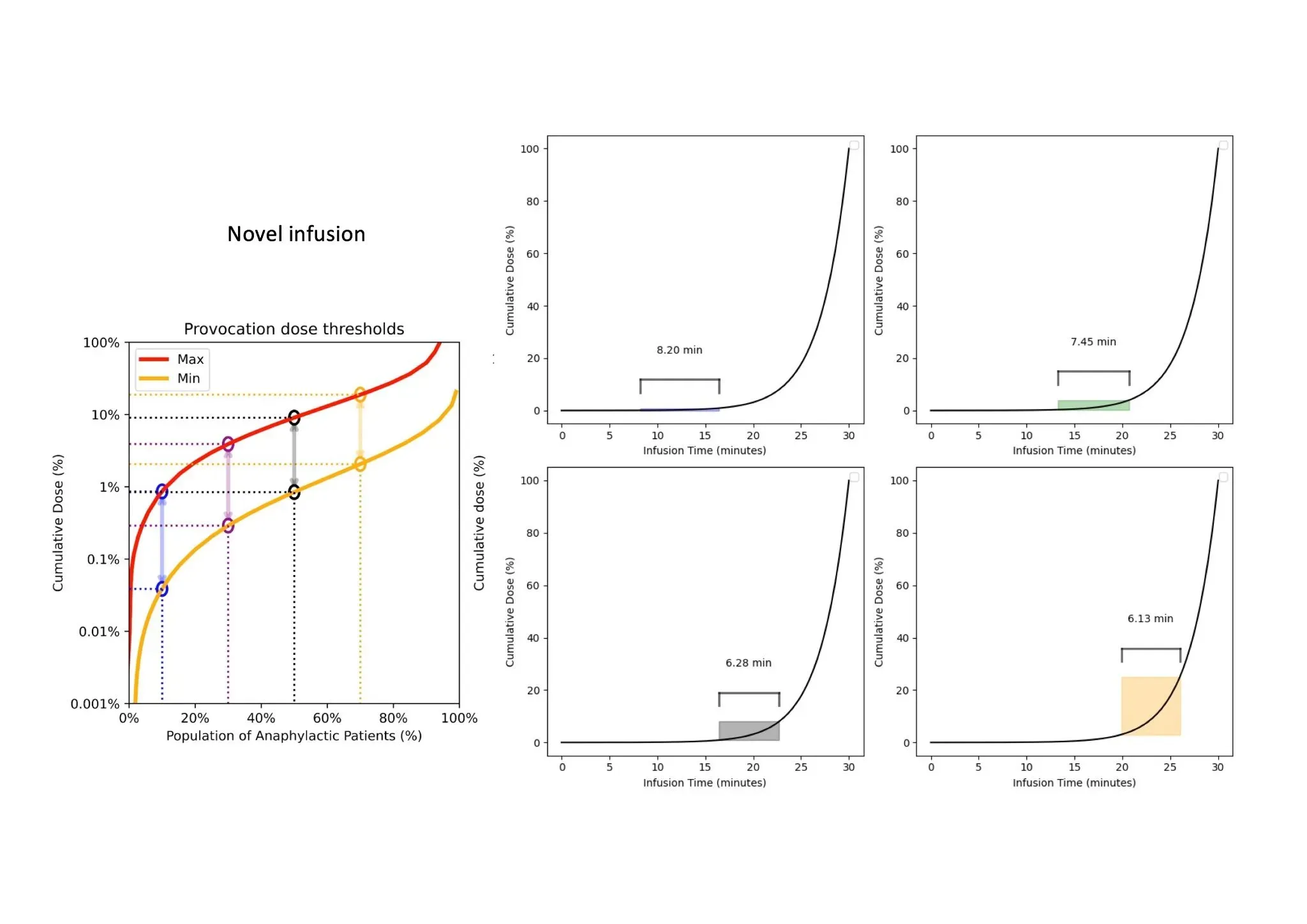
Accurate Dosing Profile
Dilution Chamber Ensures Precise, Rapid Delivery of Early Micro-doses—Overcoming Deadspace and System Compliance
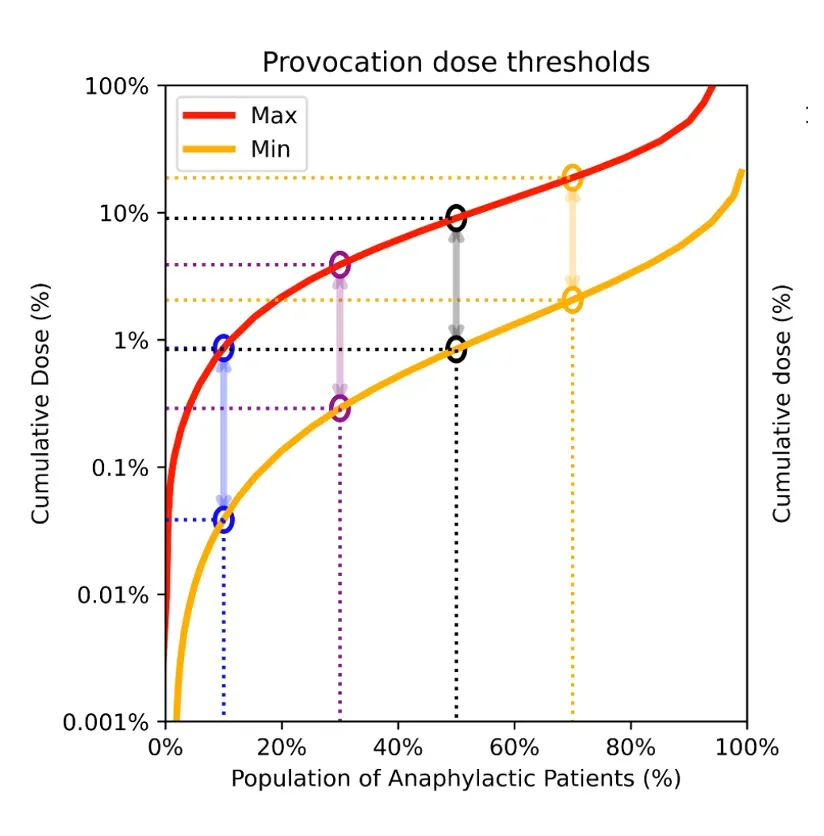
Multi-Threshold Dosing
Provides Critical Response Time Across All Sensitivity Thresholds, Allowing Rapid Detection and Intervention
Gallery


Revolutionizing care, one precise dose at a time.
Transforming Drug Anaphylaxis from Unpredictable to Preventable
Team Leader

Dr. Paul Sadleir
Chief Medical Officer
Dr. Paul Sadleir has 15 years experience in intravenous allergy diagnostics and prevention. He leads product innovation and research, focused on translating the known benefits of test dosing into a clinically appropriate solution.
Innovative Intravenous Solutions by Sadleir Laboratories
Expertise in Medical Device Innovation and Intravenous Safety
Sadleir Laboratories, based in Western Australia, is at the forefront of developing advanced infusion technologies to reduce the burden of perioperative anaphylaxis. Drawing on over a decade of clinical research and experience in perioperative hypersensitivity, the company has pioneered IRIS – the Increasing Rate Infusion System – a patented test dose syringe designed to revolutionize the safety of intravenous drug administration.
IRIS is the only device of its kind to deliver a non-linear, exponentially increasing infusion calibrated for drugs with log-normal dose-response characteristics – such as beta-lactam antibiotics and neuromuscular blocking agents – offering a safer, more responsive alternative to constant rate infusions .
IRIS: A Game-Changer in Hypersensitivity Prevention
The IRIS syringe enables clinicians to recognize allergic reactions earlier in the dosing window, reducing the total drug dose administered at the moment a reaction is detected. Unlike standard constant-rate infusions, IRIS delivers drug concentrations in a dynamic gradient, mimicking multi-step desensitization protocols within a 30-minute single-infusion format . This design significantly reduces reaction severity and facilitates safe cessation of the infusion, potentially converting life-threatening reactions into mild, self-limiting events.
The IRIS protocol has been shown in modeling to reduce administered dose by up to 12-fold at the reaction threshold compared to constant infusions . This represents a paradigm shift in intravenous drug safety and has profound implications for both challenge testing and routine drug administration.
Commitment to Patient Safety and Clinical Impact
Sadleir Laboratories’ mission is to deliver practical, high-impact innovations backed by rigorous clinical research. The IRIS system is supported by a published re-analysis of NAP6 data showing a 70% reduction in severe antibiotic anaphylaxis using test doses , and its safety and efficacy are currently being evaluated in a blinded, randomized clinical trial at Sir Charles Gairdner Hospital.
The company collaborates closely with anaesthetists, perfusionists, and academic hospitals to ensure that its technologies integrate seamlessly into existing clinical workflows. With IRIS, Sadleir Laboratories empowers clinicians with safer tools and actionable strategies to prevent one of anaesthesia’s most feared complications: intravenous anaphylaxis.